If the inner membrane is so impermeable, how do proteins enter?
The outer membrane of the mitochondria contains the protein "porin". This forms an aqueous channel through which proteins up to 10,000 daltons can pass and go into the intermembrane space. Indeed, the small molecules actually equilibrate between the outer membrane and the cytosol. However, most proteins cannot get into the matrix unless they pass through the inner membrane. This membrane contains cardiolipin which renders it virtually impermeable. This requires transport mechanisms across the membrane that are more organized and regulated. A very simple view of the process is diagrammed in this cartoon.
This figure is taken from Alberts et al, Molecular Biology of the Cell, Garland Publishing, N.Y. 1994, Third Edition
Mitochondrial import signals.

Transport across the mitochondrial membranes requires the concerted action of a number of translocation machineries. The machinery in the outer membrane is called the Tom complex (Translocator outer membrane) and that for the inner membrane is called the Tim complex (Translocator Inner Membrane). Proteins that have to go all the way to the matrix have an NH2 cleavable signal sequence (see the above cartoon).
Most proteins must be uncoiled or stretched out to go through the translocators. This involves ATP binding and is monitored and stabilized by a chaperone proteins, including hsp70. Thus, before the protein can go through Tom complex, it must become "translocation competent".
Transport through the outer membrane: characteristics of Tom complex.
Not surprisingly, the TOM complex will include import receptors that initially
recognize the signal peptide or a signal sequence (these include Tom20, Tom22, and Tom70).
Different proteins use different receptors. In the above cartoon, the receptor is
represented as a blue oval in which the signal peptide is inserted. The receptors then
bring the protein to the region containing the translocator proteins. This is actually a
complex of proteins.
It is called the General Import Pore (GIP) and it facilitates the translocation of the
presequence of the protein across the outer membrane. (the GIP is made of Tom40, Tom5, Tom
6, and Tom7). Tom40 appears to be the core element of the pore and forms oligomers.
It traverses the membrane as a series of 14 anti-parallel beta strands which form a beta
barrel. It also interacts with polypeptide chains passing through the pore.
All of the other Tom components in GIP are anchored to the outer membrane by helical
transmembrane segments (hydrophobic anchors).
Recent study of Tom40: Rapaport, D and Neupert W, Biogenesis of Tom40, Core Component of the TOM complex of mitochondria. J Cell Biol 146 321-332, 1999. Study looked at how Tom40 entered outer membrane and became a part of the GIP. The study reported that:
- First, as with many mitochondrial proteins, Tom40 requires cytosolic chaperones to prepare it for entry. In the case of this protein, becoming "translocation competent" requires ATP and a partially folded state (the latter is mediated by the cytosolic chaperone (hsp70).
- Second, when it is "competent", it interacts with the surface receptor, Tom20. There is no cleavable signal peptide however, the experiments showing the requirement for partial folding suggests targeting information is found in discontinuous sites brought together in the folded domain.
- Final insertion is into preexisting Tom complexes. This requires an intact N terminus.
- Dimerization occurs after entry into the membrane.
Characteristics of Tim Complexes
Mitochondrial proteins destined for the matrix often have a cleavable signal peptide on the protein which must be recognized before it will be admitted through the mitochondrial translocator. These proteins with "amino terminal signals" (your text), or "preproteins" or "presequences" (current literature) usually interact with Tom20 first. Then, they have to get through the outer membrane. To do that, they are transferred to the GIP complex: First, they interact with Tom22 and Tom5 which ushers them to the pore formed by Tom40. They then enter the matrix using the pore complex made of Tim23 and Tim17 which are in the inner membrane. Also, very important, their entry is dependent on membrane potential. This is set up by the electron transport complexes. Recall that hydrogen ions are being pumped into the intermembrane space creating a charge gradient that is more negative on the matrix site. This membrane potential actually helps pulls the protein into the Tim23-Tim17 channels. The protein then enters the matrix where the cleavable preprotein is clipped off by a protease, MPP. mt-hsp 70 in the matrix works with Tim44 to complete the full transfer to the matrix. mthsp70 and Tim 44 actually "pull" the protein into the matrix by a process that requires ATP. It also requires the membrane potential set up by the electron transport chain.
Some mitochondrial proteins destined for the inner membrane have a cleavable presequence followed by one or more hydrophobic membrane-spanning segments that function as stop-transfer sequences in the IM or, serve to insert the polypeptide into the IM after it gets in the matrix. These are like the Type I membrane proteins described in the unit on the rough endoplasmic reticulum.
However, other proteins do not have a cleavable targeting signal (Types II and III). Mitochondrial proteins that have an internal signal sequence (examples include a number of proteins in the inner membrane) generally interact with Tom70 as the receptor. Then, after they transit the outer membrane via the GIP complex, they enter the special Tim pathway. This may involve interactions with small Tim's of the intermembrane space and Tim22-Tim54 of the inner membrane itself.
Those proteins that do not have a cleavable targeting signal sequence often have signals with the following characteristics: They are often a stretch of positively charged amino acids (sometimes adjacent to a membrane spanning hydrophobic region). Sometimes these form loops that face the matrix. Recall the "positive inside rule" has positively charged amino acids concentrated at the cytosolic side for proteins being inserted into the rough endoplasmic reticulum. These mitochondrial proteins tend to follow this rule, only the matrix becomes the site where the positive charges are most numerous.
Examples from the literature:
Davis, AJ, Ryan, KR, and Jensen, RE Tim23p contains separate and distinct signals for targeting to mitochondria and insertion in to the inner membrane. Molecular Biology of the Cell 9: 2577-2593 (1999).
- Tim23 is one of the inner membrane translocator proteins. It does not have an amino-terminal presequence. Targeting information is found within the mature protein.
- Tim23 has 4 transmembrane segments and two positively charged loops facing the matrix. What is needed for signalling an import?
- Replaced positively charged amino acids in one or both loops with alanine residues.
- At least one of these loops is required for insertion into the inner membrane.
- The signal for targeting to mitochondria is found in at least two of the hydrophobic transmembrane segments.
Kurz, M, Martin, H, Rassow J, Pfanner, N, and Ryan, MT. Biogenesis of Tim proteins of the mitochondrial carrier import pathway: differential targeting mechanisms and crossing over with the main import pathway. Molecular Biology of the Cell 10: 2461-2474 (1999). Compared the route and binding of three Tim proteins
- Tim54 carries a amino terminal, noncleaved translocation sequence that is positively charged. However, it prefers to use Tom70 as its receptor instead of Tom20. After moving through the GIP, it uses its positively charged amino terminal sequence to enter the matrix. It required chaperones and ATP to get to the matrix.
- Tim22 is a hydrophobic protein that uses Tom20 for targeting to the OM. Then it follows the Tim route for carrier proteins, like Tim23. It does not require hsp70 or ATP for entry.
- Small Tims are normally found in the intermembrane space and are not membrane proteins. They used Tom20 for their receptor and transfer to the GIP complex. However, when Tom20 was destroyed by trypsin, leaving only Tom5, the small Tims were able to enter.
Return to Menu

The above cartoon from your text shows more ways that proteins can be inserted into inner and outer membranes, once they are recognized by the receptors. As shown by proteins in the literature examples above, the mitochondria uses both positively charged signals as well as membrane spanning hydrophobic sequences to translocate and then reach their final destination. As in the above examples, there can be multiple signal and insertion sites. However, the distribution of the charged amino acids helps orient the protein so that the positive charges are in the matrix. This is how the cytochromes in the respiratory chain or the elementary particles are inserted by mitochondrial actions.
This figure is taken from Alberts et al, Molecular Biology of the Cell, Garland Publishing, N.Y. 1994, Third Edition
The following figure is from another text by Lodish et al, Molecular Cell Biology. It shows the entire sequence of events required to take a protein into the matrix.

- Step 1: Protein unfolds as it binds to hsp70 chaperone. Red positive area indicates targeting sequence. Chaperone binding is ATP dependent.
- Step 2: Targeting sequence binds to receptor (usually Tom20)
- Step 3. Receptor ushers protein to site of translocator. Other Tom proteins involved, but Tom40 is the core of the translocator channel.
- Step 4: Protein is translocated stimulated by the membrane potential. Electron transport complexes on inner membrane have pumped H+ across to the intermembrane space, leaving the matrix more negative. This attracts the protein (the signal is positively charged). Protein moves through Tim translocators. Tim 44 and hsp70 in the matrix continue to guide and pull the protein through the pore. An ATP requiring process.
- Step 5. another chaperone (called a chaperonin), hsp60 causes the folding of the the protein into its tertiary sequence. Also an ATP requiring process.
- Step 6. Presequence is cleaved in the matrix.
What happens if an import protein is defective?
Studies of yeast have helped us learn about the receptor and translocation machinery contains a complex of proteins that work together to allow entry. In yeast, these have been named the MOMX.... series, where the number designates the protein number. An important protein in the recognition of the signal peptide and its binding to the receptor is called "MOM19". MOM 19 works with MOM 72 to recognize and bind the proteins. Then MOM22 helps the protein to pass from the receptor binding site to the insertion point at the outer membrane. The importance of MOM19 can be proved by adding antibodies to MOM19 and blocking import.
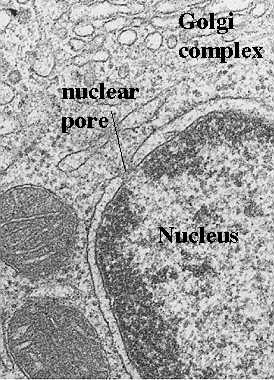
In a recent paper by Harkness et al (J Cell Biology 124: 637-648, 1995), they created mutant yeast cells that included a defective gene for MOM19.

They also included a drug resistant marker so they could selectively grow cells with the mutant gene (in the presence of the drug, p=fluorophenyl alanine, or fpa). So, the longer the cells grow in the drug, the more drug-resistant mutant cells will be found. The above electron micrographs are from their paper (cited above). They show the result of the absent MOM19 protein. What is absent in the cells grown for 16 or 32 h in the drug?
When they did the assays for the proteins, what proteins were actually missing? Tests showed that there was a dramatic decrease in most of the respiratory chain (electron transport chain) including cytochromes a/a3, and b. However, cytochrome C was unaffected. This suggests that another protein must control its import.
For more information, contact:
Gwen Childs, Ph.D.,FAAA
Professor and Chair
Department of Neurobiology and Developmental Sciences
University of Arkansas for Medical Sciences
Little Rock, AR 72205
For questions, contact this email address: